Pfizer Impfstoff
Safety over a median of 2 months was similar to that of other viral vaccines. DIE PFIZER mRNA IMPFSTOFF UNTER DEM MIKROSKOP.

Covid 19 Biontech Pfizer Impfstoff Bei Kindern Ab 12 Jahren
Efficacy shown in clinical trials in participants with or without evidence of prior infection with.

Pfizer impfstoff. Pfizer-BioNTech COVID-19 Vaccine may be effective in preventing COVID-19 in individuals 16 years of age and older and the known and potential benefits of the Pfizer-BioNTech COVID-19 Vaccine. Food and Drug Administration FDA approval on August 23 2021 for individuals 16 years of age and older. Vaccine Corona Pfizer Corona-Impfstoff von Biontech.
Once vaccines are approved by the FDA companies can market the vaccines under brand names. Address reprint requests to Dr. Learn about SARS-CoV-2 the virus that causes COVID-19.
An additional dose may be given to people with a severely weakened immune system at least 28 days after their second dose. A booster dose may be given at least 6 months after the second dose for people aged 18 years and older. 14122020 pfizer which produced the vaccine alongside german company biontech began shipping the doses from its michigan warehouse sunday directly to.
Ein deutscher Forscher nahm eine Probe des Pfizer-Impfstoffs und untersuchte sie mit einem Standardmikroskop mit einer hellen Mikroskoplampe und nahm sie durch das Mikroskop auf Video auf das Video ist 11 Minuten lang und wurdeContinue reading de. COVID-19 vaccination Baada ya chanjo ya Pfizer Comirnaty ya COVID-19 After your Pfizer vaccine Swahili - Kiswahili. Acteur majeur dans les biosimilaires en oncologie recommandation vaccinale.
The vaccine has been known as the Pfizer-BioNTech COVID-19 Vaccine and will now be marketed as Comirnaty for the prevention of COVID-19 disease in individuals 16 years of age and older. VAERS DL calculation. COVID-19 mRNA Pfizer- BioNTech vaccine analysis print Reaction Name Total Fatal Blood disorders Anaemia deficiencies Anaemia folate deficiency 1 0 Anaemia vitamin B12 deficiency 3 0.
Absalon at Pfizer 401 N. Middletown Rd Pearl River NY 10965 or at email protected. In countries where the vaccine has not been approved by the relevant regulatory authority it is an investigational drug and its safety and efficacy have not been established.
In Pfizers briefing document submitted to an FDA advisory committee meeting held on 10 December 2020 to discuss Pfizers application for emergency use authorisation of its covid-19 vaccine the company made no mention of problems at the Ventavia site. COMIRNATY is the brand name for the Pfizer-BioNTech COVID-19 Vaccine. Developing the Pfizer-BioNTech COVID-19 vaccine is only possible through the dedicated work of thousands of individuals and those who volunteer to take part in research.
Last 6 month COVID deaths assuming 90 death reduction 91868 90 82681 lives saved per 360M doses -826813600001000000 -230 DM. Home Pfizer Biontech - BiontechPfizer Impfstoff zugelassen. COVID-19 vaccination உஙகளடய கவட-19 தடபபசய நஙகள இடடககணட பறக Pfizer பஃபஸர After your Pfizer vaccine Tamil - தமழ.
Conclusion With up to 6 months of follow-up and despite a gradually declining trend in vaccine efficacy BNT162b2 had a favorable safety profile and was highly efficacious in preventing COVID-19. Your immune system cells then recognise the spike protein as a threat and begin building an immune response against it. Global Information About PfizerBioNTech COVID19 Vaccine also known as BNT162b2 The approval status of the PfizerBioNTech COVID19 Vaccine varies worldwide.
Pfizer-BioNTech COMIRNATY Name Change. Category Deaths per M doses Est lives saved per M doses last 6 mo -230 Excess deaths per M VAERS Pfizer only 458. This type of vaccine uses a genetic code called RNA to make your bodys cells produce the coronavirus specific spike protein.
Pfizer is a messenger RNA mRNA vaccine. We are grateful to all of our clinical trial investigators and their study teams who are partnering with us in this effort and to all of the participants and their families who have and will volunteer to help make a difference. Der Impfstoff von Biontech und Pfizer wurde von Ende Juli bis Mitte November in einer Studie mit insgesamt 44820 Probanden untersucht.
And our scientific efforts that we hope will help bring an end to the current global health crisis. The vaccines have been developed on mRNA technology and have received temporary approval in. The study published in the New England Journal of Medicine 1 found that vaccine efficacy between the first and second doses was 52 95 credible interval 295 to 684 with 39 cases of covid-19 in the vaccine group.
Two COVID-19 vaccines Comirnaty from BioNTecPfizer and Moderna are used in Norway. A two-dose regimen of BNT162b2 conferred 95 protection against Covid-19 in persons 16 years of age or older. Pfizer-BioNTech COMIRNATY received US.
Seite 3 von 3 kbv steckbrief impfstoff comirnaty von biontechpfizer 19. 9 2020 New York-based Pfizer and the German company BioNTech made history by announcing that their coronavirus vaccine had an efficacy rate. Funded by BioNTech and Pfizer.
Comirnaty is given as two injections usually into the muscle of the upper arm 3 weeks apart. 4582302 per 6 month efficacy period. In South Africa where the SARS-CoV-2 variant of concern B1351 beta was predominant 100 95 CI 535 1000 VE was observed.
What you can do to stay safe and prevent the spread. In countries where the vaccine has not been approved by the. While we continue to see the devastating impact of the coronavirus pandemic around the world we are committed to helping keep people safe and informed.
The Pfizer-BioNTech COVID-19 Vaccine is a suspension for intramuscular injection administered as a series of two doses 03 mL each 3 weeks apart. The Pfizer and BioNTech covid-19 vaccine may provide some early protection starting 12 days after the first dose the peer reviewed results of a phase III trial have found. The next day the FDA issued the authorisation of the vaccine.
49 6131 9084-0 For questions on general topics.

Pfizer Covid Jab Can Be Stored In Fridge For A Month Eu Regulator
Corona Impfstoff Biontech Pfizer Beantragen Notzulassung In Usa Br24
Vom Lastwagen In Die Altenheime Verteilung Des Impfstoffs Zdfheute
Corona Impfungen Bei Kindern Biontech Pfizer Starten Tests Zdfheute

Biontech Pfizer Us Zulassung Fur Covid Impfstoff Beantragt

Biontech Pfizer Corona Covid 19 Impfstoff Am 22 01 2021 In Meerbusch Biontech Pfizer Corona Covid 19
Us Zulassung Von Biontech Pfizer Impfstoff Erwartet Wissen Umwelt Dw 09 12 2020
Coronavirus Pandemie Usa Lassen Vakzin Fur Auffrischungen Zu Tagesschau De
Pfizer Biontech Covid 19 Vaccine Wikipedia

Cominarty Biontech Pfizer Impfstoff Mit Aufgezogener Spritze Mit Einer Impfstoffflasche Cominarty
Peter Liese Fda Recommends Emergency Approval For Biontech Pfizer Vaccine For 5 11 Year Old Children
Over Half Of German Respondents In Favor Of Ending Vaccination Prioritization Newsroom Universitat Hamburg
Corona Impfung In Osterreich Mit Impfstoff Von Biontech Pfizer Corona Vaccination In Austria With Vaccine From Biontech Pfizer Stock Photo Alamy
Corona Impfung In Osterreich Mit Impfstoff Von Biontech Pfizer Corona Vaccination In Austria With Vaccine From Biontech Pfizer Stock Photo Alamy

Corona Pandemie Wohl Deutlich Mehr Impfstoff Ab 2 Quartal Tagesschau De

Cominarty Biontech Pfizer Impfstoff Mit Aufgezogener Spritze Kochsalzloesung Und Aufkleber Comina

Coronavirus Germany Hopeful Of Earlier Eu Vaccine Approval News Dw 15 12 2020
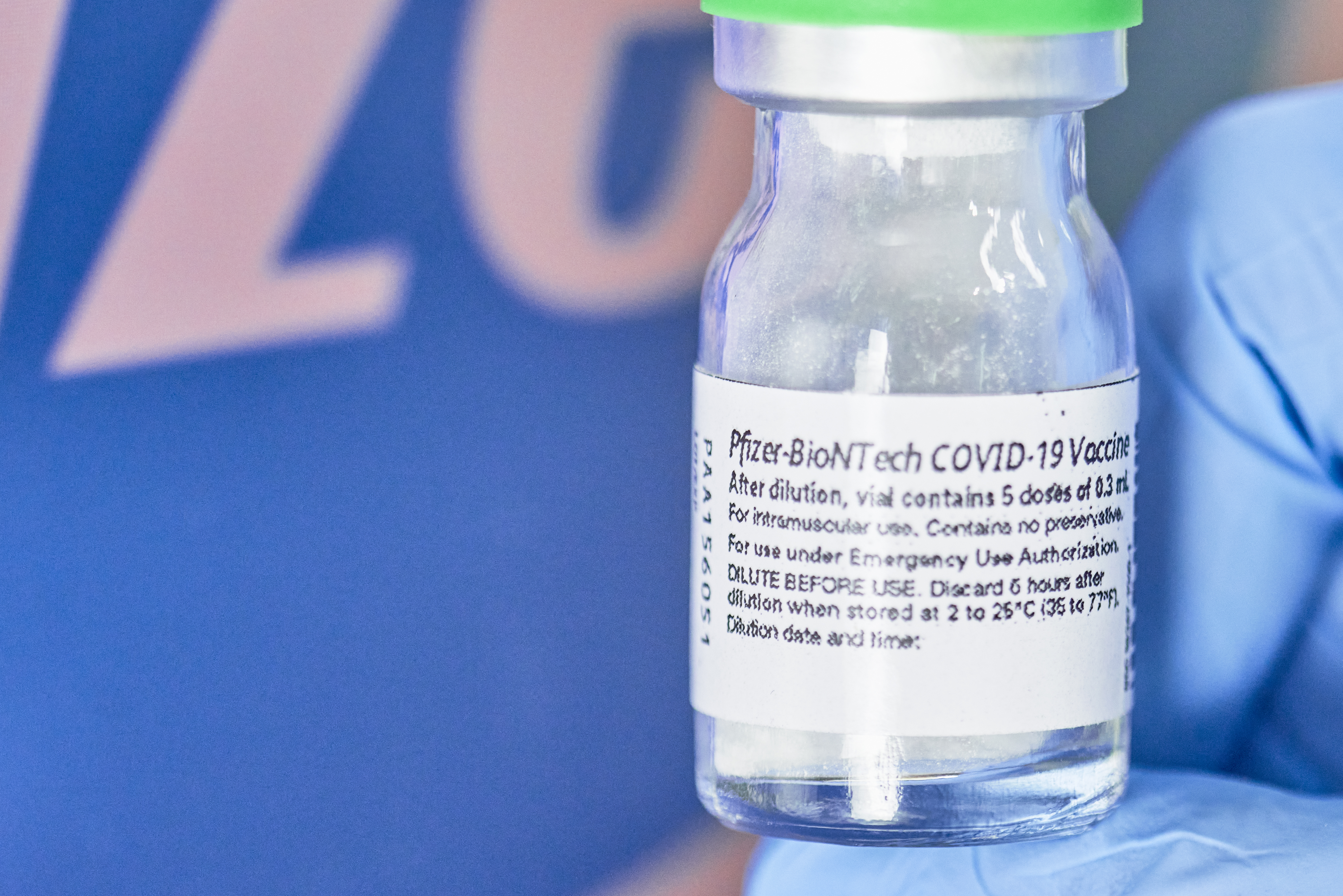
File Pfizer Biontech Impfstoff Ampulle 50744958791 Jpg Wikimedia Commons

Professors Health Officials React To Pfizer Fda Approval News Thebatt Com